In the opening to We Have Never Been Modern (1993) sociologist Bruno Latour describes a newspaper report on the hole in the ozone layer. The article links a dizzying array of items: the Antarctic, refrigerators, aerosols, the arcane chemistry of inert gases, international scientific collaborations, and the design of factory assembly lines. Together they weave “the most esoteric sciences and sordid politics.” Ozone is a classic Latourian science-society hybrid: it resists all efforts to fence off pure from applied science and science from politics.
One Latourian hybrid now fascinating researchers and the public is the gene-editing technique CRISPR-Cas9. What began with an attempt to build a better yogurt now has journalists speculating about Brave New World (1932) scenarios. Scientists, ethicists, entrepreneurs, and officials across the globe want to reap the benefits while guarding against errors and side effects.
Because humans are not simply genetic robots, CRISPR probably can't do more than cure certain rare diseases.
So, yogurt. In 2007 scientists working for Danisco, a Danish food ingredient company now owned by Dupont, invented a method for encouraging virus resistance in Streptococcus thermophilus, a bacterium critical to yogurt and cheese production. The Danisco researchers took advantage of earlier observations that bacterial DNA often contains strangely arranged sequences that run first in one direction (e.g., from base A to base Z), then are separated by an apparently random spacer sequence, followed by the same sequence in reverse order (from base Z to base A). It did not take long to realize that the spacer sequences matched regions of DNA in bacteriophages, the class of viruses that attack bacteria. These patterns, which are widespread in the bacterial world, became known as clustered regularly interspaced short palindromic repeats, or CRISPR. The researchers invented their method to change the resistance to viral infection of S. thermophilus by tinkering with the viral spacer DNA in the CRISPR sequence.
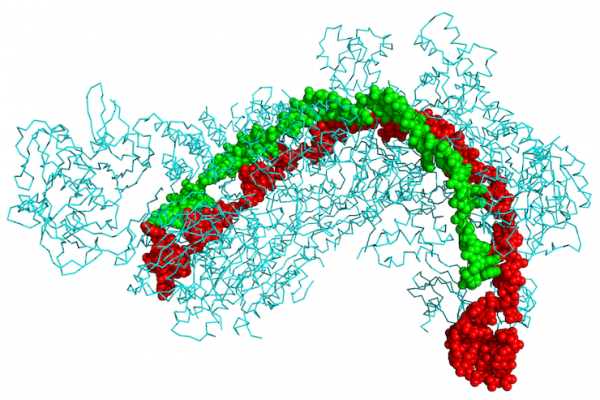
The next step came from biologists Jennifer Doudna and Emanuelle Charpentier (Nobel watchers, keep your eye on these two), who found that CRISPR sequences bind to a protein called Cas9, which holds the palindromic DNA stretches in place, making the spacer available for precise cutting. The system—which also involves some specialized RNA molecules—acquired the name CRISPR-Cas9, and in no time flat CRISPR-Cas9 DNA editing kits appeared on the commercial market, making the technique widely available. That was 2012. By January of the following year, research teams had used CRISPR-Cas9 to add, delete, suppress, or activate targeted genes in mice, fruit flies, fish, worms, plant crops, and, yes, human cells.
Although the process represents a huge increase in editing accuracy, it suffers from what researchers call “off-target effects” or, in lay terms, unintended consequences.
CRISPR-Cas9 is now the biologist’s fastest, most sequence-specific technique for the rewriting of genetic information. The process can be used to fix genetic information, if it is deemed broken. And CRISPR-Cas9 can be used to change when and where that information is read out in the cell as an RNA molecule. Beyond making stronger yogurt and cheese bacteria, some scientists hope it can be used to correct the products of debilitating genetic mutations, such as sickle cell anemia, and to treat arthritis and cancer. Like many a good Latourian hybrid, CRISPR-Cas9 is now embroiled in a U.S. patent battle over who owns the intellectual rights to what may become a highly remunerative technology.
With all this potential come serious challenges. Although the process represents a huge increase in editing accuracy, it suffers from what researchers call “off-target effects” or, in lay terms, unintended consequences. On-target edits also can result in the unforeseen. For example, unbeknownst to the experimenter, a targeted gene might act in several different tissues, not just the one designated for repair.
To address such concerns, this year the Innovative Genomics Initiative at the University of California, Berkeley sponsored a Forum on Bioethics with the goal of identifying problems with the potential use of CRISPR-Cas9 in humans. Attendees made several recommendations: continue discussions with bioethicists, scientists, and the wider public about the development and ethical use of CRISPR-Cas9; make research evaluating the safety and efficacy of CRISPR-Cas9 genetic engineering rigorous and transparent; and convene a globally representative, multidisciplinary conference to discuss the scientific, medical, legal, and ethical issues raised by the technique. Finally, the Berkeley conference recommended that scientists and governments strongly discourage efforts to modify human sperm or eggs—in essence any efforts to make eugenic changes in humans.
Possible eugenic deployment of CRISPR-Cas9 has raised the loudest alarms.
Possible eugenic deployment of CRISPR-Cas9 has raised the loudest alarms. If in fact modification of individual genes can be causally linked to desired changes in mature bodies, then, in principle, CRISPR-Cas9 eugenics wouldn’t be that hard. Widespread in vitro fertilization techniques already make one-cell human embryos accessible outside the body. To change the DNA in an entire organism, including future eggs or sperm, just add the CRISPR-Cas9 cocktail to the fertilized egg before embryonic cell division starts. Indeed, this year Chinese scientists tried to modify the human beta-globin gene (HBB), which in mutated form causes the genetic disease beta-thalassemia. The scientists used eighty-six human embryos donated for research by couples at an in vitro fertilization clinic. But the attempt was far from successful. Fifteen of the embryos died, and of the remaining seventy-one, fifty-four were tested for the intended genetic changes. Among these, only four had the changes. They also showed the dreaded off-target effects, meaning that there were potentially dangerous mutations in genes other than HBB.
Despite the poor results, the paper provoked a good deal of anxiety. The National Academy of Sciences responded by organizing an international summit scheduled for this fall as well as an international committee charged with conducting “a comprehensive study of the scientific underpinnings and clinical, ethical, legal, and social implications of human gene editing.” Some hope that a research moratorium—akin to the 1975 pause to evaluate the safety of genetically modified organisms and 1997’s halt in research on human reproductive cloning—will give everyone time to catch their breath and work out standards and rules of ethical practice before proceeding more deliberately.
Others argue that we need to dig deeper. The standard framing for ethics debates is to weigh the promise of future medical benefits against the medical and ethical risks of a new treatment. But in a conference held in Atlanta this past May, one group of bioethicists instead pushed a social justice angle. Who, they wanted to know, might receive treatment? Who is left out of decision-making? Who profits from the new techniques? And does treatment increase or decrease inequality? These social justice ethicists point out that some of the most severe medical challenges originate from poverty, pollution, and environmental disasters. “Let’s progress with cures,” they write, while also getting smart about “prevention, access, affordability.”
If expectations for CRISPR-Cas9 prove overheated, it may be because we keep insisting on a false model, as if humans are just the expressions of a genetic code established once and for all at conception.
Meanwhile, some fear that CRISPR-Cas9 opens the door to a eugenic dystopia of the kind dramatized in the 1997 film Gattaca. Others, myself included, suspect we will be saved from that fate by the actual biology of the matter. Because humans are not simply genetic robots, because genetic networks, environment, and development work together in tangled ways, CRISPR-Cas9 probably can’t do more than cure certain single-locus genetic diseases. That is very well, but such diseases are rare. Most genetic effects on human phenotype come from tens to hundreds of genes involved in complex interactions with each other and with the environment.
As useful as CRISPR-Cas9 has proven in research, we should be wary of bio-hype. We’ve seen enough of that to know skepticism is warranted. The war on cancer began forty-four years ago, but cancer remains one of the major causes of death in the United States. The Human Genome Project, for its part, did not find the expected strong genotype-phenotype connections: the clear DNA “blueprint” supposedly underlying traits of individuals. And genome-wide association projects have turned up a confusing jumble of interconnections: many genes with small effects rather than a few variations making obvious contributions to disease.
If expectations for CRISPR-Cas9 prove overheated, it may be because we keep insisting on a false model of development in which humans are just the expressions of a genetic code established once and for all at conception. But one can’t accurately read—or write—the future in genes alone. We will know more of CRISPR-Cas9’s true potential in a few years. But in the meantime, there is no doubt the technology is scientifically and socially an important development, which deserves our attention as its story evolves.